 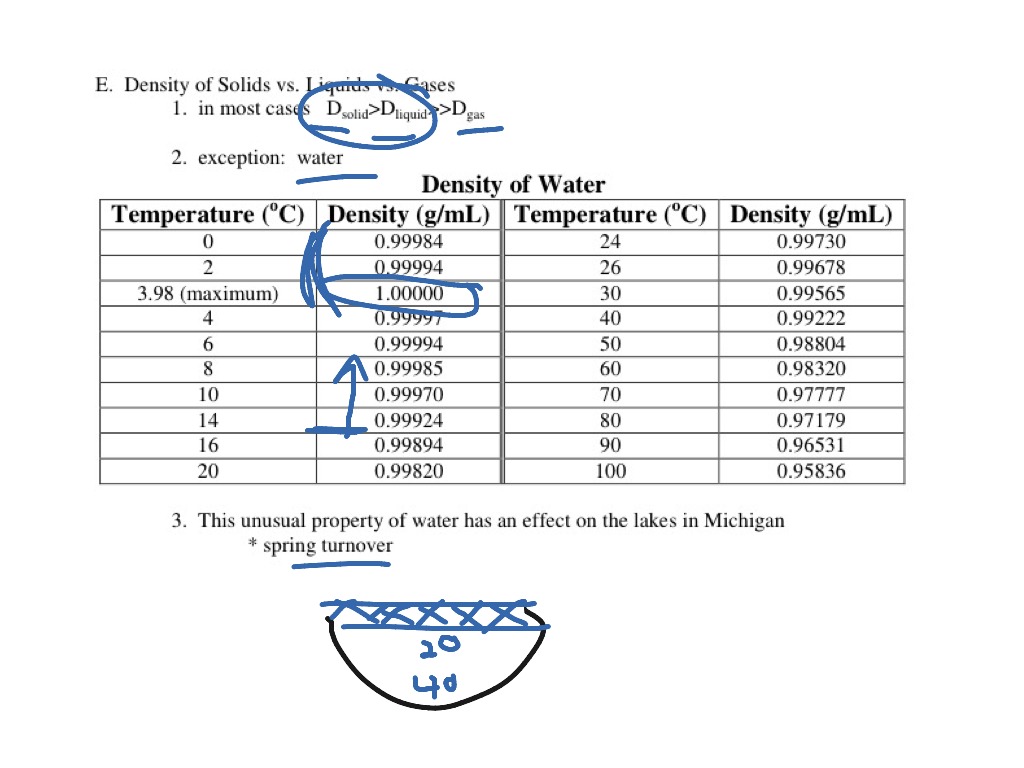
A hydrometer has a weighted bulb and a cylindrical stem with lines on it that represent how far the weighted bulb sinks in the liquid. The simplest way to find the density of a liquid is with a hydrometer. How To Calculate The Density Of A Liquid? Therefore, heavy ice sinks in normal water and floats in heavy water. When a neutron is added to an atom of hydrogen, it makes deuterium 10.6% denser than your normal water. Deuterium is an isotope of hydrogen in which there is one electron, one proton, and one neutron. Ice of heavy water sinks in water owing to its higher density because deuterium atoms replace hydrogen items in heavy water. The reason for this unusual negative thermal expansion is because of orientation-dependent, and strong, intermolecular forces, and they’re usually seen in molten silica. If you cool it down, even more, it expands and becomes less dense.Normally, when you cool a liquid from room temperature, it becomes denser, but with pure water, it reaches its maximum density when it is approximately at 4☌.This relation is unimodal and non-linear in nature. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
